Medtech
There is a lack of evidence to demonstrate the effective use of continuous glucose monitors (CGMs) in people not living with diabetes (PNLD), a study led by researchers at UCL and Birmingham Children’s Hospital has found. The researchers concluded there is currently little published evidence on how accurate CGMs are in measuring blood glucose levels [...]
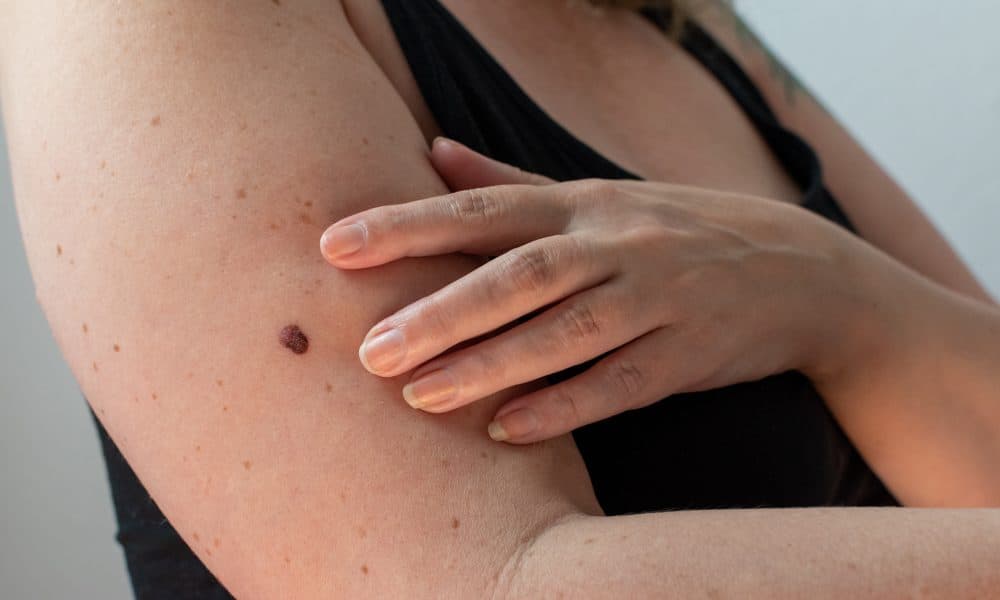
An AI-powered medical device has successfully identified five percent of all UK melanoma cases in the month of May as part of a Post-Market Clinical Follow-up (PMCF). AUTODERM is a research-backed Software as a Medical Device (SaMD) incorporated in the myGP app. The clinical research-backed AI dermatology analytics engine is CE marked and registered with [...]
Researchers in the US are developing a new range of wound care technologies, including smart bandages that would automatically sense and respond to changing conditions inside a wound. The high-tech dressings would provide continuous data on healing and potential complications, including infections or abnormal inflammation, and could deliver medications or other treatments in real time. [...]
A new project is underway in Lewisham, London to develop the educational resources needed to train healthcare professionals on how to use novel AI-powered results to diagnose respiratory illnesses. The project will focus on the N-Tidal device, developed by medtech company TidalSense, which uses AI to improve the speed and accessibility of Chronic Obstructive Pulmonary Disease [...]
Researchers in the US have developed a field-ready screening tool for traumatic brain injury (TBI). The Advanced Military Measure of Olfaction (AMMO) kit includes an array of scents, deployable anywhere from the battlefield to the football field, to help screen for TBIs in minutes. The kit was developed by a team at Southwest Research Institute [...]
Scientists at the Broad Institute of MIT and Harvard have improved a gene-editing technology that is now capable of inserting or substituting entire genes in the genome in human cells efficiently enough to be potentially useful for therapeutic applications. The advance, from the lab of Broad core institute member David Liu, could one day help [...]
A new, wafer-thin patch has been developed that incorporates a flexible electronic circuit, a gel made from tapioca starch and gelatin, as well as a friendly bacteria that can help treat skin conditions. For years, Professor Bozhi Tian’s lab has been learning how to integrate the world of electronics—rigid, metallic, bulky—with the world of the [...]
NHS patients in England are set to benefit from faster access to innovative and cutting-edge medical technologies under new plans. Proposals set out in a joint NHS England and NICE consultation outline a new route for MedTech developers to access NHS funding to fast-track clinically and cost-effective products for use by the healthcare system. The plans will [...]
A world-first minimally invasive brain pacemaker is now available on the NHS for drug-resistant epilepsy patients across the UK. Over 600,000 people in the UK have focal epilepsy. Although most patients take at least one drug to treat the symptoms, around 30 per cent of epilepsy patients are drug resistant, meaning they continue to suffer [...]
The Medicines and Healthcare products Regulatory Agency (MHRA) has published a proposed framework for international recognition of medical devices to ensure access to MedTech innovations. The MHRA’s statement of policy intent describes how the UK Government intends to recognise regulatory approvals from Australia, Canada, the European Union and the United States of America, depending on [...]