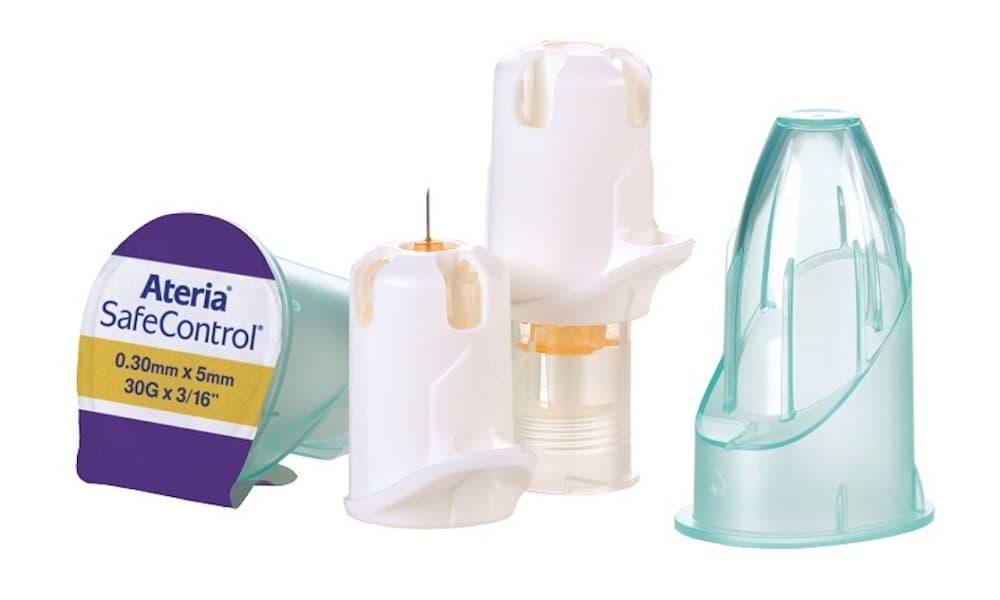
Oxfordshire-based Owen Mumford, a medical device manufacturer, has been appointed as the sole supplier for insulin
Ateria SafeControl safety pen needles will now be used in all NHS Wales hospital environments, as well as by community nurses who also have access to the new procurement framework.
The device is also listed on the Drug Tariff, following its approval for use by the NHS Prescription Services, meaning GP practices are now able to prescribe the device and pharmacists can dispense to suitable patients throughout the UK.
Covering all health boards and trusts in Wales, the Ateria SafeControl active safety pen needle was chosen for the four-year framework agreement following a competitive tender process.
Paul Lee, medical device training manager at Swansea Bay University Health Board, said: “When the sharps regulation came into force, we found that devices were not ready – they were cumbersome and cost prohibitive.
“The trial demonstrated that Ateria SafeControl delivered the same functionality as passive safety pen needles. Also important was the ability to visually see the entry site of the needle which helps to ensure correct dose delivery, while enabling the HCP to use the same injection technique as a conventional pen needle.
“Going forward we predict there will be a reduction in needle stick injuries as a result of NHS Wales’ adoption of safety engineered devices.”
Leanne Adam, marketing manager at Owen Mumford, added: “The cost of NSIs is considerable and far reaching. NHS employees may need time off work, which puts pressures on teams and can have a significant impact on the wellbeing of the individual.
“There is a financial impact too where compensation has to be paid. A single needlestick injury is one injury too many. These should be avoided where possible.”