The National Institute for Health and Care Excellence (NICE) has published draft guidelines recommending that flash glucose monitoring or continuous glucose monitoring be made available for all adults with type 1 diabetes in the UK.
The guideline also suggests that all children living with type 1 diabetes should use continuous glucose monitoring.
Diabetes UK said the guidelines are a hugely-welcome step towards more people having access to the life-changing technology, which improves blood sugar control and makes life easier for people with type 1 diabetes, their parents and their carers.
Chris Askew OBE, chief executive at Diabetes UK, said: “We are pleased to see such great strides being made in ensuring more people with type 1 diabetes have access to flash monitors on the NHS.
“This technology transforms the lives of those using it, improving both their quality of life and their diabetes self-management.
“We also know that, for those who do have access to flash monitors, it has been particularly beneficial while face-to-face diabetes care has been understandably limited during the restrictions of the COVID-19 pandemic.
“We look forward to continuing to work with NHS England and others to ensure those who can benefit from this life-changing technology have access to it”.
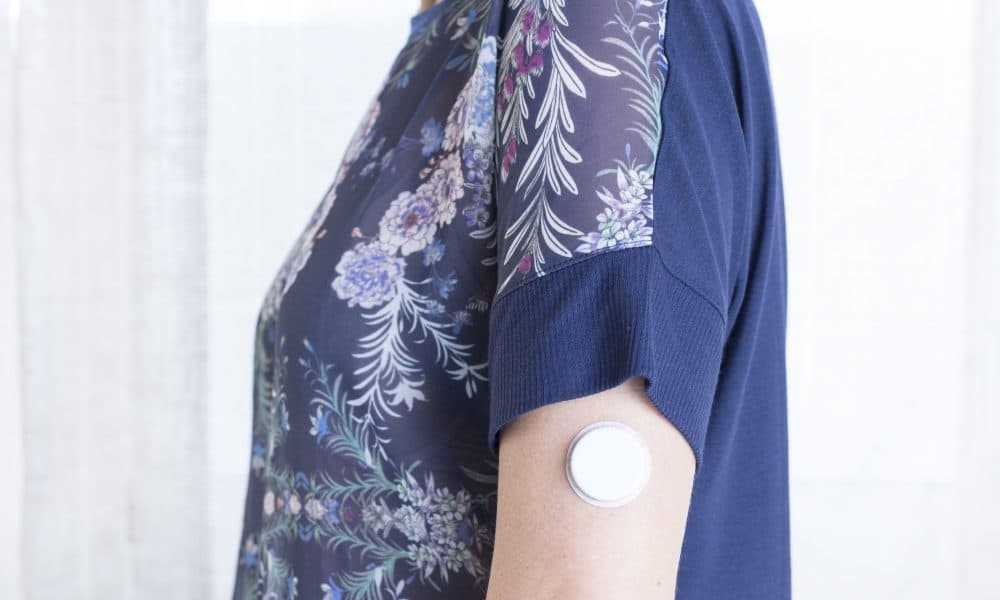
Flash monitors have a sensor that easily attaches to the back of the arm, allowing patients to check their glucose quickly and easily with a simple one-second scan.
The wearable gadget links to an easy-to-use app on your phone, where patients can access the data gathered by the device.
Unlike conventional blood glucose monitors they allow patients to view patterns over time, not only showing current and previous levels but also where they’re headed.
The draft NICE proposals also recommend that people with type 2 diabetes who use insulin intensive therapy (four or more injections a day) and experience regular hypoglycaemia or severe hypoglycaemia also be given access to Flash.
Diabetes UK said it is pleased to see NICE recognise that more people living with type 2 diabetes can benefit from Flash, something it has highlighted consistently.
Chris Askew said: “At the moment, NICE only recommends access to CGM in very limited circumstances and the guidelines don’t mention Flash at all.
“We’ve been involved every step of the way in this guideline update process, pushing for better access to this important technology. We’re thrilled to see NICE listen to us, and others, who have been calling for them to recommend wider Flash and CGM use.
“While health care professionals don’t have to follow NICE guidelines, such as those proposed today, they are influential. Health care professionals frequently use them to inform the decisions they make for their patients’ care.
“We’ll be responding to the consultation on this draft guideline and welcome this big step forward. We expect the final guidance to be published in March 2022.
“We look forward to continuing to work with NICE, the NHS and local Integrated Care Systems (the NHS partnerships created to join up health and care services available to patients locally).
“We sincerely hope that, when the final guidelines are published, local leaders within healthcare systems will do all they can to make them a reality.”
Eligible patients in the UK are currently able to access flash monitors on prescription from their local GP or diabetes team, helping them to better manage their blood sugar levels.
Amanda Pritchard, NHS England’s chief executive, said: “Flash glucose monitoring is a great example of where technology and digital solutions can help individuals to live more independent lives, better manage their own conditions, and avoid more acute health problems developing.
“It is a testament to the hard work of NHS colleagues working across diabetes care that we have managed to roll out these devices to half of Type 1 patients and that the real-world data is helping to inform potential future service development in this area”.