
A new breakthrough in brain-computer interfaces (BCIs) could pave the way for wireless devices that improve life quality and independence for people with paralysis.
BCIs are an emerging assistive technology, enabling people with paralysis to type on computer screens or manipulate robotic prostheses just by thinking about moving their own bodies.
For years, investigational BCIs used in clinical trials have required cables to connect the sensing array in the brain to computers that decode the signals and use them to drive external devices.
Now, for the first time, scientists have demonstrated use of an intracortical wireless BCI with an external wireless transmitter. The study involved clinical trial participants with tetraplegia – partial or total loss of movement of all four limbs and torso.
The system is capable of transmitting brain signals at single-neuron resolution and in full broadband fidelity without physically tethering the user to a decoding system.
The traditional cables are replaced by a small transmitter about two inches in its largest dimension and weighing a little over 1.5 ounces. The unit sits on top of a user’s head and connects to an electrode array within the brain’s motor cortex using the same port used by wired systems.
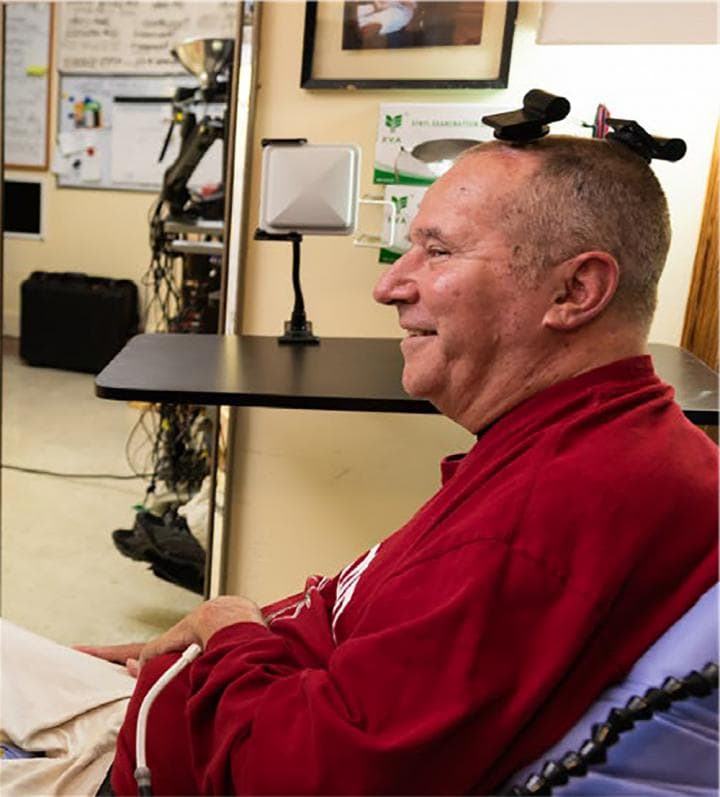 For the study, published in IEEE Transactions on Biomedical Engineering, two clinical trial participants with paralysis used the BrainGate system with a wireless transmitter to point, click and type on a standard tablet computer.
For the study, published in IEEE Transactions on Biomedical Engineering, two clinical trial participants with paralysis used the BrainGate system with a wireless transmitter to point, click and type on a standard tablet computer.
The study showed that the wireless system transmitted signals with virtually the same fidelity as wired systems, and participants achieved similar point-and-click accuracy and typing speeds.
John Simeral, an assistant professor of engineering at Brown University, a member of the BrainGate research consortium and the study’s lead author, said: “We’ve demonstrated that this wireless system is functionally equivalent to the wired systems that have been the gold standard in BCI performance for years.
“The signals are recorded and transmitted with appropriately similar fidelity, which means we can use the same decoding algorithms we used with wired equipment. The only difference is that people no longer need to be physically tethered to our equipment, which opens up new possibilities in terms of how the system can be used.”
The researchers say the study represents an early but important step toward a major objective in BCI research: a fully implantable intracortical system that aids in restoring independence for people who have lost the ability to move.
While wireless devices with lower bandwidth have been reported previously, this is the first device to transmit the full spectrum of signals recorded by an intracortical sensor.
A high-broadband wireless signal enables clinical research and basic human neuroscience that is much more difficult to perform with wired BCIs.
The new study demonstrated some of those new possibilities. The trial participants – two men aged 35 and 63 respectively – were both paralysed by spinal cord injuries.
They were able to use the system in their homes, as opposed to the lab setting where most BCI research takes place.
Unencumbered by cables, the participants were able to use the BCI continuously for up to 24 hours, giving the researchers long-duration data including while participants slept.
Leigh Hochberg, an engineering professor at Brown, said: “We want to understand how neural signals evolve over time.
“With this system, we’re able to look at brain activity, at home, over long periods in a way that was nearly impossible before. This will help us to design decoding algorithms that provide for the seamless, intuitive, reliable restoration of communication and mobility for people with paralysis.”
The device used in the study was first developed at Brown in the lab of Arto Nurmikko, a professor in Brown’s School of Engineering.
It was designed to transmit high-fidelity signals while drawing minimal power. In the current study, two devices used together recorded neural signals at 48 megabits per second from 200 electrodes with a battery life of over 36 hours.
While the device has been used successfully for several years in basic neuroscience research, additional testing and regulatory permission were required prior to using the system in the BrainGate trial. Nurmikko says the step to human use marks a key moment in the development of BCI technology:
“I am privileged to be part of a team pushing the frontiers of brain-machine interfaces for human use. Importantly, the wireless technology described in our paper has helped us to gain crucial insight for the road ahead in pursuit of next generation of neurotechnologies, such as fully implanted high-density wireless electronic interfaces for the brain.”
The BrainGate consortium is an interdisciplinary group of researchers from Brown, Stanford and Case Western Reserve universities, as well as the Providence Veterans Affairs Medical Center and Massachusetts General Hospital.
In 2012, the team published landmark research in which clinical trial participants were able, for the first time, to operate multidimensional robotic prosthetics using a BCI.
That work has been followed by a steady stream of refinements to the system, as well as new clinical breakthroughs that have enabled people to type on computers, use tablet apps and even move their own paralysed limbs.
Study co-author Sharlene Flesher, who was a postdoctoral fellow at Stanford and is now a hardware engineer at Apple, says: “The evolution of intracortical BCIs from requiring a wire cable to instead using a miniature wireless transmitter is a major step toward functional use of fully implanted, high-performance neural interfaces.
“As the field heads toward reducing transmitted bandwidth while preserving the accuracy of assistive device control, this study may be one of few that captures the full breadth of cortical signals for extended periods of time, including during practical BCI use.”










