Europe
The Medicines and Healthcare products Regulatory Agency (MHRA) has published a proposed framework for international recognition of medical devices to ensure access to MedTech innovations. The MHRA’s statement of policy intent describes how the UK Government intends to recognise regulatory approvals from Australia, Canada, the European Union and the United States of America, depending on [...]
An MHRA and NICE partnership, funded by Wellcome, is addressing key challenges for regulating and evaluating digital mental health technologies (DMHTs) with input from patients, the public and mental health professionals. The long-term objective of the three-year project, which began in 2023, is to formulate guidance and other sources of information for developers, healthcare professionals, [...]
Messages sent to millions of patients via the NHS App have saved the NHS £1.1 million on the cost of previously sending the information via text message. During 2023/24, 22.5 million messages were sent through the NHS App, which would have previously been sent as a text message or letter. The average cost of sending [...]
The creation of a first-of-its-kind imaging system at the University of Surrey could help the UK lead a revolution in medicines and other materials sciences. The new facility promises a better understanding of the effects of pharmaceuticals and could eventually lead to better drug development. Thanks to a £3 million grant from the Engineering and [...]
The NHS is set to roll out the first ever targeted treatment for brain tumours in children and young people, which can be taken at home and has been shown to significantly slow the progression of the disease. Dabrafenib with trametinib has been found to stop the disease progressing for more than three times as [...]
Prothea Technologies, a spin-out company from the Universities of Bath and Edinburgh, has launched with a mission to improve lung cancer diagnosis and treatment. The firm, which has attracted €12m in funding, seeks to develop a medical device to diagnose lung cancer lesions using a combined endoscope and image-processing system capable of examining the molecular [...]
Pixyl.Neuro, a generative AI software for brain MRI analysis has received FDA approval. The US is now one of 12 countries where the platform has been approved by regulators. French medtech company Pixyl has announced FDA 510(k) clearance for its Pixyl.Neuro software, a next-generation AI software for brain MRI analysis. the platform recently demonstrated enhanced [...]
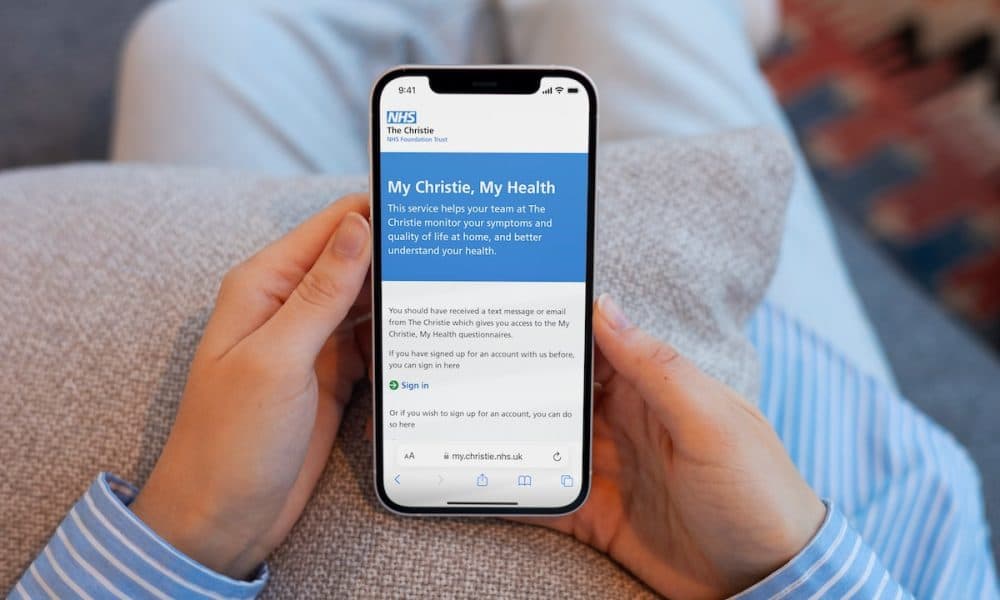
The Christie NHS Foundation Trust has signed a two-year contract with Avenue3 to build and deploy its Patient Engagement Platform and E-Referral solutions, which are underpinned by openEHR technology, making it the first trust in England to do so. The project is set to support greater digital functionality for clinicians providing care and improved patient [...]
NHS trusts are exploring new opportunities to use infrastructure that they already own. The aim? To better engage with patients, alleviate their anxieties, and inform patients what is happening regarding their care, whilst at the same time reducing pressures on busy wards. Matt O’Donovan, Chief Executive at SPARK TSL, explains. What do patients want to [...]
Sharon Lamb, Partner, Bella North, Senior Associate and Michaela Novakova, Associate, McDermott Will & Emery